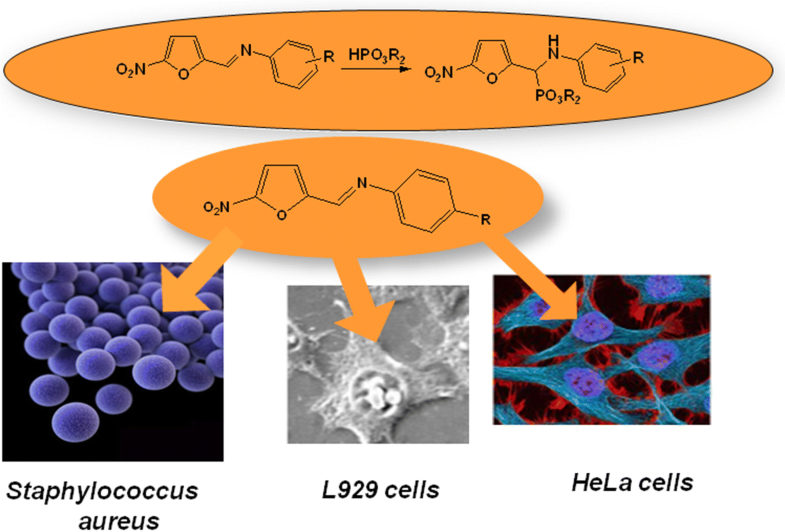
Antibacterial action of (5-nitrofurfuryl)-derived aminophosphonates and their parent iminesJarosław Lewkowski, Marta Morawska, and Aleksandra Kowalczyk University of Łódź, Łódź, Poland
E-mail: jaroslaw.lewkowski@chemia.uni.lodz.pl Abstract: Ten aminophosphonates bearing 2-nitrofuran moiety and two N-aryl 5-nitro-furfuralaldimines, namely, N-(2-nitrofurfurylidene)-p-toluidine and N-(2-nitrofurfurylidene)-p-anisidine, were tested in aspect of their antibacterial action. O,O′-diphenyl derivatives were found inactive, while O,O′-dimethyl and O,O′-diethyl derivatives were revealed to act moderately efficiently against clinical isolates of S. aureus, especially against methicillin-resistant (MRSA) strains. A high activity against these strains was found for N-(4-methylphenyl)-5-nitrofurfuralaldimine and N-(4-methoxyphenyl)-5-nitro-furfuralaldimine, but they showed the cytotoxicity at a dangerous level. Keywords: 5-Nitrofurfural-derived aminophosphonates ; S. aureus ; Antibacterial activity ; MRSA strains ; Cytotoxicity Full paper is available at www.springerlink.com. DOI: 10.1007/s11696-018-0597-1
Chemical Papers 73 (2) 365–374 (2019) |
Monday, April 27, 2026 |
|||
© 2026 Chemical Papers |
||||